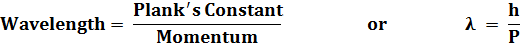
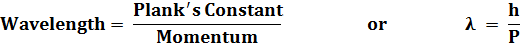
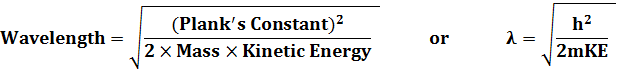
To begin to understand and experiment with objects with masses greater than that of an electron, run the simulation of the experiment for electrons.Throughout this experiment keep the same energy. Next choose pions and repeat the process. Repeat for neutrons, and finally protons. (A pion has a mass 270 times that of the electron while the neutron and proton masses are about 2,000 times that of the electron.) After doing these experiments, feel free to vary any of the parameters except the energy. Then, answer the following questions. Compare the diffraction patterns made using the other types of matter with that of the electron.